Trade shows
Company Updates

Genabio passed ISO 13485 Certification
2022-05-27
Genabio passed ISO 13485 Certification

Genabio delivered millions of INDICAID Antigen Rapid Test Kits to USA
2022-01-13

Genabio delivered millions of Babio Virus Transport Kit to USA
2022-01-03

Genabio delivered more than 5000 oxygen concentrators to Indian
2021-06-05

Genabio received CE Certificate
2021-04-13
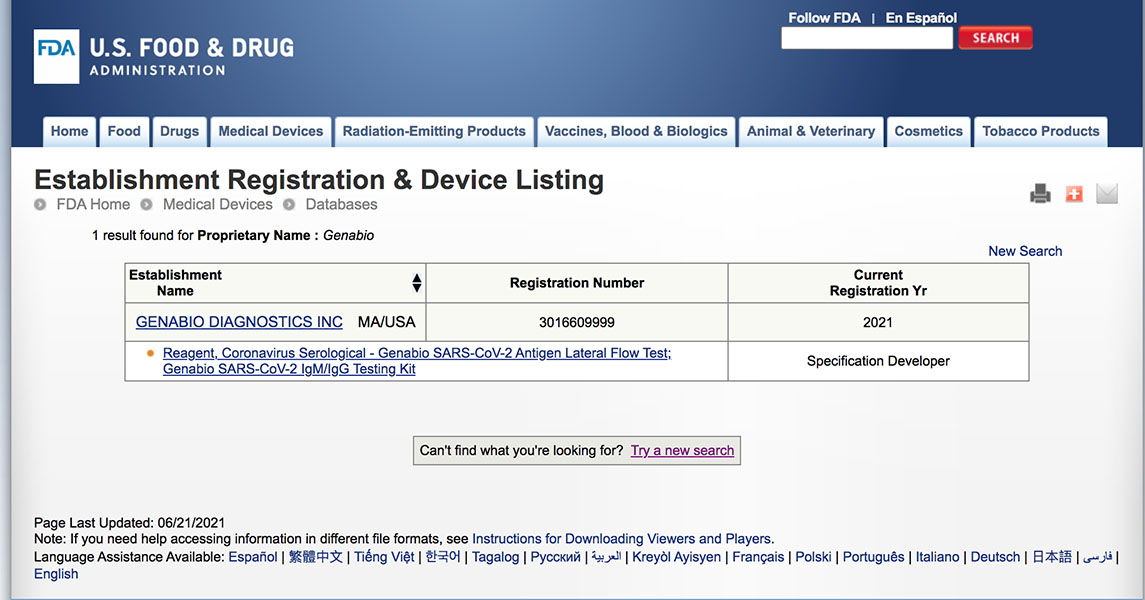
Genabio add into FDA Emergency Notification Listing
2021-02-08

Genabio delivered more than 60 million Swabs to US
2020-12-30

Genabio received CE Certificate
2020-11-13

Genabio received CE Certificate
2020-10-29

Genabio delivered more than 2 million Universal Sample Kits to Canada
2020-08-12
Subscription
We can help you figure out your needs.




